

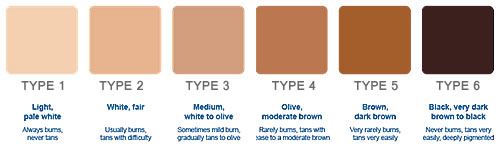
Observation of skin over 35 years has shown me that the original burn time that a skin could tolerate when young becomes shorter with age, along with less tolerance to heat. Older skin also displays a greater predisposition to post inflammatory hyperpigmentation after an injury, even if in the lower Photo type scale. The colour tone of skin is divided into 6 categories as an assessment of the response of skin to sun exposure. This is known as the Photo type scale, or Fitzpatrick Skin Type and was first developed in 1975.
Later in the 1990’s the Fitzpatrick Skin Type Scale was updated and although the Scale was a good reference for the skins burn time, it did not give any indication of the skins ability to accumulate melanin and change colour (tan).
Nor did it indicate the risk for skin cancer or post inflammatory hyperpigmentation, however this scale is used as a point reference for this discussion.

Why does skin burn time decrease with age?
The turnover and regeneration of cells change as skin ages. Based on the strength of genetic heritage these changes accelerate and compound the amount of oxidative stress each skin cell is exposed to by the living and working environment of the individual.Cells have different stem cell resources and regeneration cycles, for example, the melanocyte cell has no stem cell resource and very few regeneration cycles, most of which are completed by the age of eighteen.The melanocyte resides in the basal layer resting on the lamina lucida of the dermojunction and is considered the 5th in line of epidermal barrier defence. A dendritic cell, the melanocyte works in synergy with the ascending keratinocyte in the spinosum layer by passing melanin carrying melanosomes. So there are two cells involved in the melanogenesis of an ageing skin.
Here are some considerations of why skin burn time decreases with age.
1. No stem cell resource to draw on as melanocytes age, and documented that skin loses a minimum of 10% functioning melanocytes every 10 years from the age of 35.
2. The remaining melanocytes have little if any regeneration cycles.
3. Mitochondrial DNA ageing (MDNA) and Cellular Senescence of both melanocytes and keratinocytes mean inefficient cellular function and epidermal barrier defence formation.
4. MDNA ageing or Senescence Melanocyteshave a reduced production of melanosomes and poor quality and quantity in the percentage of melanin granules produced. This reduced production may be due to incorrect endoplasmic reticulum folding during the melanosome/melanin formation.
5. Cellular antioxidant defence systems decline with age, causing greater intracellular oxidation, cellular stress, and inflammation.
6. Dendrites become shorter as the melanocytes age, making it difficult to place melanosomes and pigment evenly throughout the spinosum layer, thereby reducing the epidermal barrier defence.
7. Essential fatty acid deficiency due to poor absorption of nutrients is common among ageing clients andcontributes to the malfunctioning dendrites of melanocytes and keratinocyte plasma membranes.
8. Any changes to the strength and function of the keratinocyte reduce all levels of epidermal barrier defence such as cell envelope formation, corneocyte compaction and resulting stratum corneum density.
9. Calcium levels required for corneocyte compaction decline with age, affecting stratum corneum density and strength.
10. Atrophy of the spinosum layer is also a contributing factor in the inefficient placement of melanin.
Conclusion:
1. Discuss all of the above considerations with your ageing patients or clients, perhaps even include some information in your next newsletter.
2. Discuss with the patient or client the importance of increasing the SPF factor of sun protection product to be higher than previously used and to use protective clothing such as broad brim hats, long sleeves dresses or shirts, longer skirts, and pants or leggings.
3. Include antioxidant protection for application under the sun protection product and after sun exposure.
4. Offer Environmental Defense facials over the summer period.


